How to probe a western blot
3:47
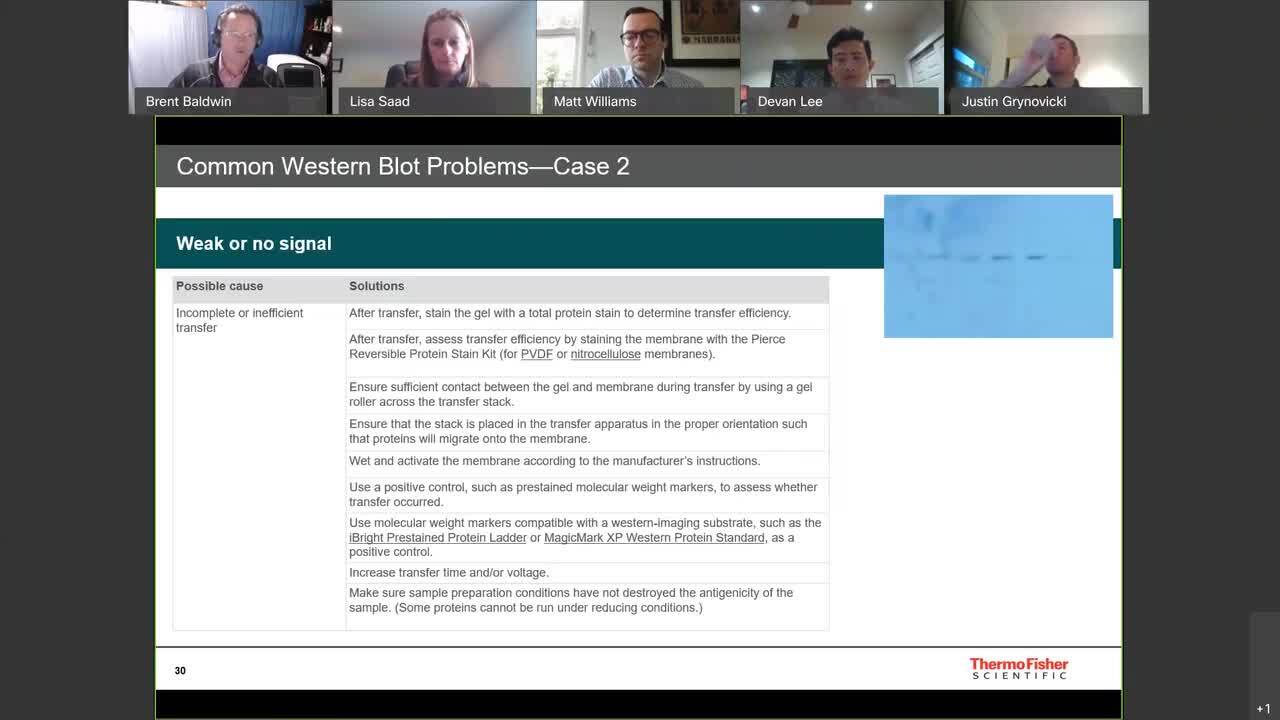
"After Western blot transfer, the membrane is ready to be probed for your target protein of interest, or the antigen. The detection process involves blocking the membrane and probing with primary and secondary antibodies. The blocking step is critical—it entails blocking the membrane surface with a protein or another molecule that is nonreactive to the antibody that will be used to probe for your target protein or antigen. Nitrocellulose and PVDF membranes have a high protein binding affinity which is essential for successful protein transfer. The blocking step ensures that the antibody binds specifically to the antigen rather than nonspecifically to the membrane surface. Nonspecific binding can result in a high background signal that prevents detection of your protein of interest. Blocking buffers are often comprised of tris- or phosphate-buffered saline with Tween-20 detergent, and up to 5% of a nonreactive protein such as bovine serum albumin, nonfat milk, casein, or gelatin. There are also protein-free blocking buffers that eliminate problems associated with traditional protein-based blockers, such as cross-reactivity and interference from glycosylated proteins. The exact formulation of blocking buffer and the incubation period depend on the membrane type, the antibody, and the substrate system used. Identifying the formulation that yields the highest signal-to-noise ratio requires empirical testing. To block your membrane, cover the membrane with blocking buffer and incubate for 30-60 minutes at room temperature with gentle agitation. For typical incubation trays, use at least 15 mL for mini blots and 30 mL for midi blots to fully cover the membrane. Avoid low volumes, as differences in agitation and coverage can produce high or uneven background. Now, the blocked membrane is ready to be probed with the primary antibody. The primary antibody should be diluted in the blocking buffer per supplier recommendations with or without detergent. Incubate the membrane with gentle agitation for 1 hour at room temperature or overnight at 4 degrees Celsius. Remove any unbound primary antibody by performing a series of washes. The typical wash buffer is tbs- or pbs-based with 0.05-0.1 percent detergent. Wash the membrane 3 times for 10 minutes each in wash buffer with gentle agitation. Next, dilute the secondary antibody in wash buffer or blocking buffer. The final concentration of the secondary antibody will depend on the detection system used. Incubate the membrane for 1 hour at room temperature with gentle agitation. Remove any unbound secondary antibody by performing a series of washes. Wash the membrane 6 times for 5 minutes each in wash buffer with gentle agitation. Washing at this step is important to help limit background signal. The membrane is now ready for detection. Find out more information about buffers for western blotting at thermofisher.com/western buffers "