-
Play video Single-use bioreactor scaling calculator demo
Single-use bioreactor scaling calculator demo
Learn how to use a single-use bioreactor scale-up calculator to simplify complex bioprocess calculations, improve accuracy, and support efficient development and production workflows.
8:35
-
Play video Aegis Film for DynaDrive Single-Use Bioreactors
Aegis Film for DynaDrive Single-Use Bioreactors
Pair DynaDrive single-use bioreactors with Aegis film to simplify validation and support global compliance. PFAS- and Ergifos-168–free, it delivers strength and compatibility from media prep through production.
1:02
-
Play video CHO Cell Line Development with CHOvantage GS Kit
CHO Cell Line Development with CHOvantage GS Kit
Develop high-titer CHO cell lines faster with the CHOvantage GS Cell Line Development Kit. Achieve >7 g/L titers, stable clones in 14 weeks, and scalable workflows with transposon-based technology.
1:27
-
Play video The Upstream Advantage: Unlocking bioprocessing potential
The Upstream Advantage: Unlocking bioprocessing potential
Hear from Thermo Fisher Scientific colleagues and learn about their perspectives on the future of bioprocessing.
3:26
-
Play video Gibco Efficient Pro Feed 3 Protein Production Workflow
Gibco Efficient Pro Feed 3 Protein Production Workflow
Boost CHO-K1 GS titers by up to 146% with Efficient-Pro Feed 3 and Feed Enhancer—a two-part, chemically defined, animal-origin-free system designed with advanced metabolomics to enhance productivity and maintain critical product quality.
1:41
-
Play video How to operate a Thermo Scientific DynaDrive 5L Single-Use Bioreactor
How to operate a Thermo Scientific DynaDrive 5L Single-Use Bioreactor
Watch a walkthrough of how to properly operate a Thermo Scientific DynaDrive 5L Single-Use Bioreactor.
8:28
-
Play video The Upstream Advantage: Unlocking bioprocessing potential
The Upstream Advantage: Unlocking bioprocessing potential
Listen to various Thermo Fisher Scientific colleagues share their insights on the future of bioprocessing.
8:00
-
Play video Affinity Chromatography: Innovative Approach for Enhanced Yield and Purity in Plasma-derived IgG
Affinity Chromatography: Innovative Approach for Enhanced Yield and Purity in Plasma-derived IgG
A robust IVIG production method using direct adsorption & affinity chromatography enhances IgG yield, purity, and subclass retention, reduces impurities, enables resin reuse for cost savings & removes ethanol precipitation for optimized processing.
31:23
-
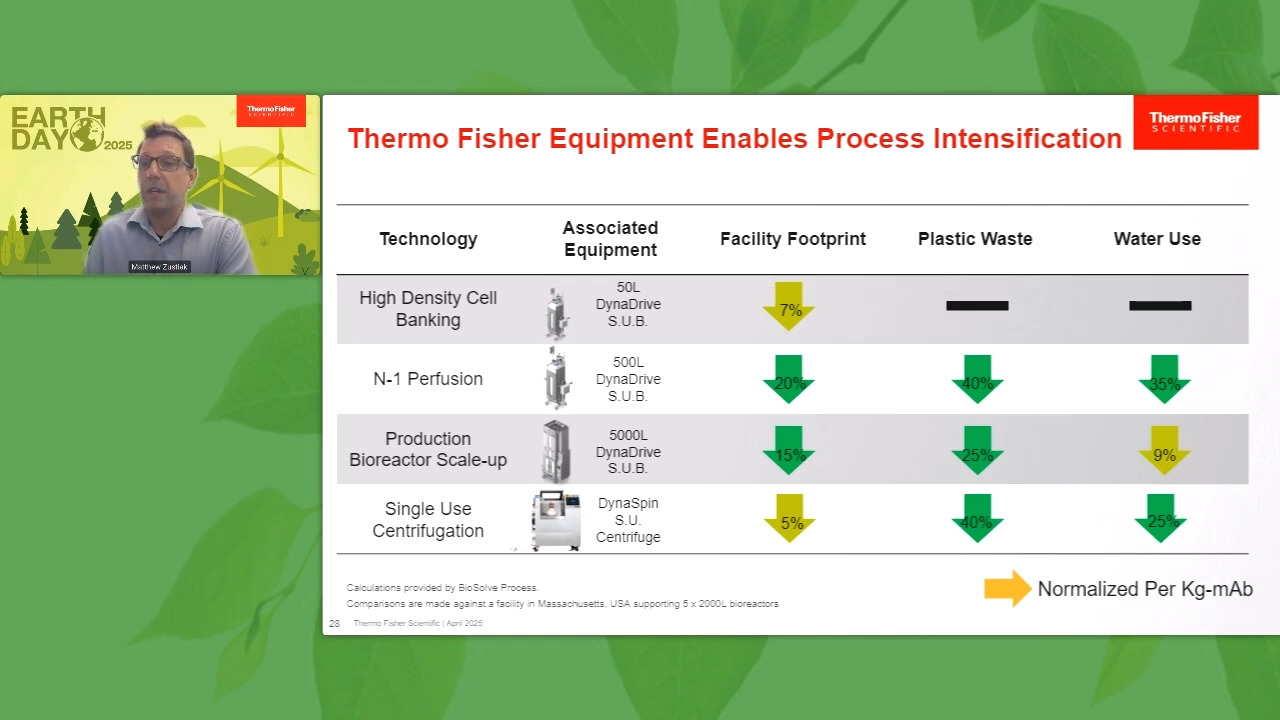
Play video Sustainable Manufacturing through Efficient Bioproduction Unit Operations
Sustainable Manufacturing through Efficient Bioproduction Unit Operations
Ready to discover how to make bioprocessing greener and more cost-effective? Learn about key environmental and economic impacts, the urgency of managing plastic waste, and a three-pronged strategy to address this growing industry challenge.
34:24
-
Play video Navigating global regulatory landscapes in an evolving biopharma industry
Navigating global regulatory landscapes in an evolving biopharma industry
Join our webinar for expert insights, engaging discussions, and interactive Q&A sessions. Discover the latest trends, expand your knowledge, and connect with professionals in your field. Don’t miss this chance to enhance your skills and stay ahead!
27:52
-
Play video E-Gel Power Snap Plus Cinematic
E-Gel Power Snap Plus Cinematic
The Invitrogen E-Gel Power Snap Plus Electrophoresis System is a compact, precast agarose gel running and imaging station.
0:39
-
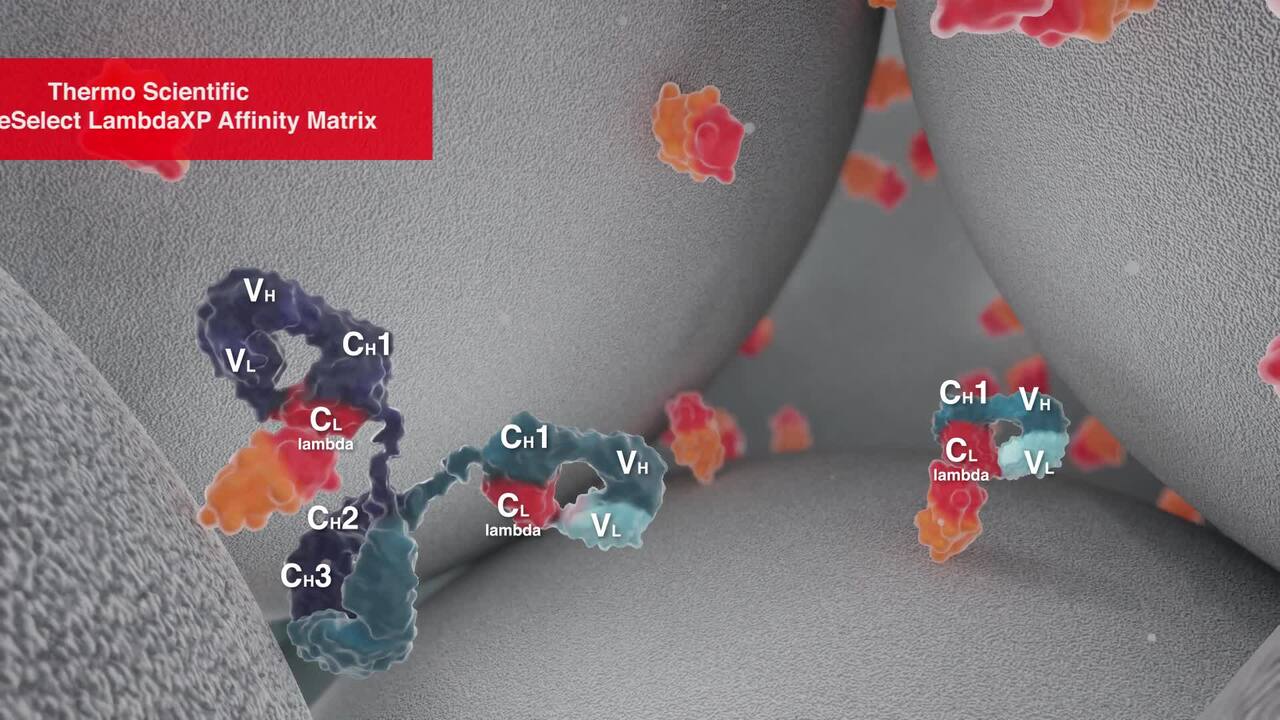
Play video CaptureSelect Affinity Antibody Resins Cinematic
CaptureSelect Affinity Antibody Resins Cinematic
CaptureSelect® products possess a combination of unique properties, including selectivity, affinity, and stability.
1:01
- Load More
Shopping Tool
eSolutions
Thermo Fisher Scientific
Note: You clicked on an external link, which has been disabled in order to keep your shopping session open.
Ok
Share this video
Embed
Size: x pixels