Step-by-step guide to successful western blot analysis
3:05
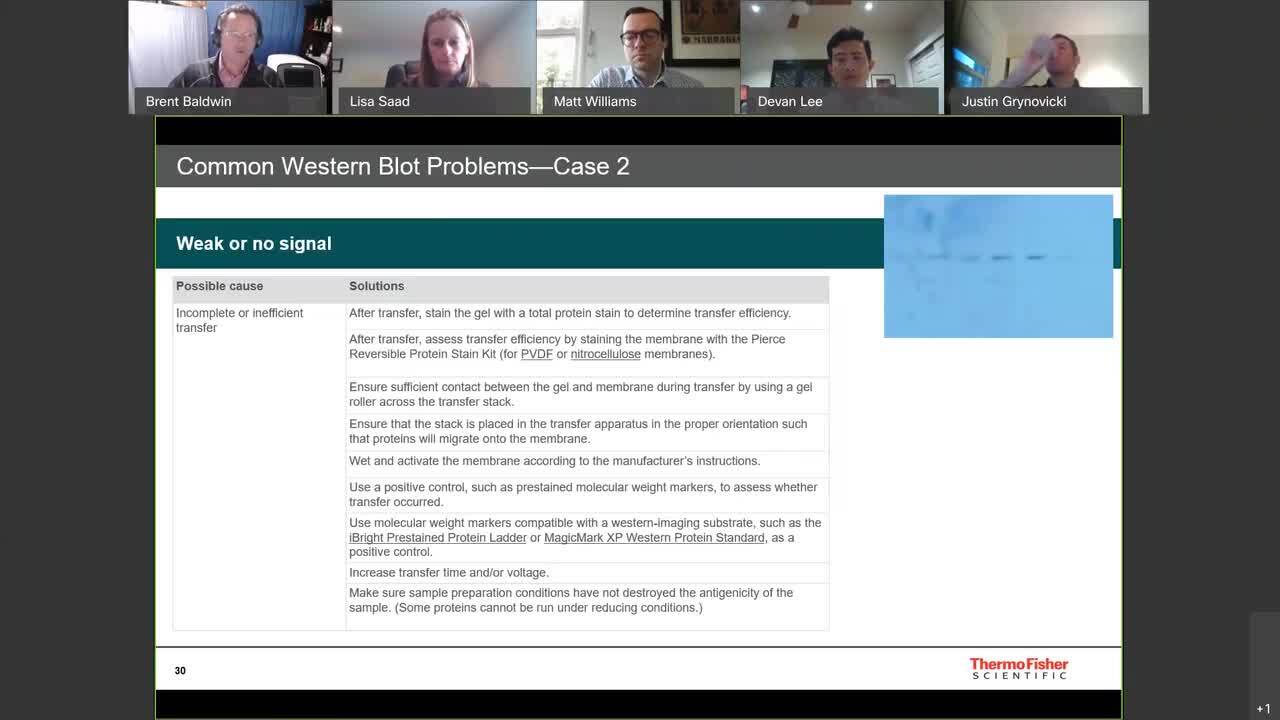
Western Blotting is the most widely used technique for protein detection. In this video, we will cover the 3 major steps in generating a western blot: Separate, Transfer, and Detect. After protein samples have been prepared, the mixture is separated by molecular weight using protein gel electrophoresis, which is a standard laboratory technique by which charged protein molecules are transported through a matrix by an electrical field. Once the proteins have been separated in the gel, they must be transferred to a solid support membrane, which facilitates protein detection through the use of antibodies specific to the target protein. Using a traditional apparatus, the protein transfer process can take 30 to 60 minutes to complete. However, new technologies have been developed that can cut the transfer time down to as little as 7 minutes. Once complete, the efficiency of the transfer can be confirmed by staining the membrane with a reversible stain. The membrane is now ready for detection. This is a multi-step process, and the first step is minimizing nonspecific binding by using a blocking agent. To do this the membrane should first be incubated with a protein containing solution such as nonfat dry milk or a purified protein like bovine serum albumin, or BSA. Proteins in this blocking solution occupy the empty spaces on the membrane to help prevent detection antibodies in the next step from sticking to the membrane. Recent advancements in blocking agents have helped make this step faster and more efficient. In this step, the membrane is incubated with the primary antibody specific to the target to allow the antibody to bind its target. Any unbound primary antibody is washed away. A secondary antibody that is specific to the primary antibody is then added. The secondary antibody is typically tagged with a reporter enzyme like Horse Radish Peroxidase or a fluorophore. As before, any unbound secondary antibody is washed away. If using an enzyme-based system, a few different substrates can be used, which produce either a colorimetric or chemiluminescence signal. Chemiluminescence detection is the method of choice in many protein laboratories, as it provides high sensitivity and convenience for detection with a film or digital imaging equipment. Today more researchers are taking advantage of fluorescently conjugated secondary antibodies that eliminate the need for substrates and provides the ability to detect multiple proteins at the same time. Western blot imaging and documentation can be performed using an X-ray film for chemiluminescence detection, or increasingly with western blot imaging systems equipped with charge-coupled device cameras. In summary, the western workflow consists of 3 major steps: 1. Separate proteins by gel electrophoresis 2. Transfer proteins from the gel to a solid support and 3. Detect, where we use antibodies specific to the target protein to visualize the protein of interest Depending on your experimental needs, there are reagents and equipment that can help optimize every step of the western blot process. Find out more about western blotting at thermofisher.com/western.